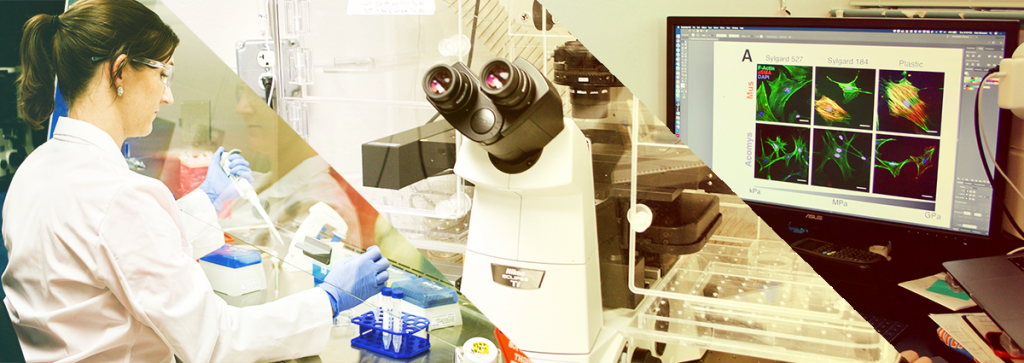
Dr. Chelsey Simmons, Assistant Professor, Department of Mechanical & Aerospace Engineering and Affiliate faculty of the J. Crayton Pruitt Family Department of Biomedical Engineering, at the UF Herbert Wertheim College of Engineering has been awarded a $1.8 million grant by the National Institutes of Health to study models of scar-free healing in mammals. The goal of Dr. Simmons’ research is to develop engineering models that can be used in medical studies of fibrosis (scarring) in human/animal injury and disease. The grant is a Maximizing Investigators Research Award (MIRA), given by the National Institute of General Medical Sciences division of NIH. The grant provides flexibility in use of the funds to allow the researchers to pursue promising results from initial projects.
Discovery of a mammalian model
Scientists know a lot about regeneration in lower vertebrates – starfish, salamanders, etc. – but that knowledge hasn’t translated well to human health because of the dramatic differences between humans and starfish. A few years ago, though, biologists studying the taxonomy of wild animals in the deserts of Northeast Africa came across a small mammal that seemed truly miraculous. Dr. Malcom Maden, Professor of Biology at UF, and his team characterized the African spiny mouse as the only mammal known to regenerate. Its ability to regenerate skin has been of particular interest to Dr. Maden and to Dr. Simmons. Even mice observed to have relatively large skin wounds due to predation in the wild seemed to heal perfectly. All tissues associated with the skin – the epidermis, dermis, sweat glands, etc. – grew back without scarring.
An engineering approach to a biological issue
Traditionally, when studying cells, researchers have cultured them on plastic petri dishes or glass slides. These relatively stiff culture surfaces activate fibroblasts (cells involved in fibrosis) into scar-making mode before researchers can study them at baseline. Taking a mechanical engineering approach, Dr. Simmons and her team asked the question, “How can we make the cell environment outside the body (in vitro) more like the cell environment inside the body (in vivo) to study inactivated, ‘normal’ fibroblasts?” Their solution was to design a “softer” culture surface by using hydrogels. In the “Model Systems of Mammalian Regeneration” project, the team hopes to view the spiny mouse fibroblasts at rest and then determine the exact steps that occur in their activation and study how they heal wounds without scarring.
“By studying the cells outside the body but in a more native (biomimetic) environment, we hope to develop models for scar-free healing that can be applied to medical research,” Simmons said. “This work could progress in so many ways, since farm animals and pets often share diseases affected by fibrosis like scarring, cancer, and autoimmune disease. I have collaborators across UF’s Colleges of Medicine, Veterinary Medicine, and Liberal Arts and Sciences, and we are poised to tackle many different problems.” Simmons views the collaborative atmosphere between UF colleges as a big plus in her research: “At UF, the medical research community genuinely appreciates engineering and understands what engineers can offer in terms of new approaches to problems.”
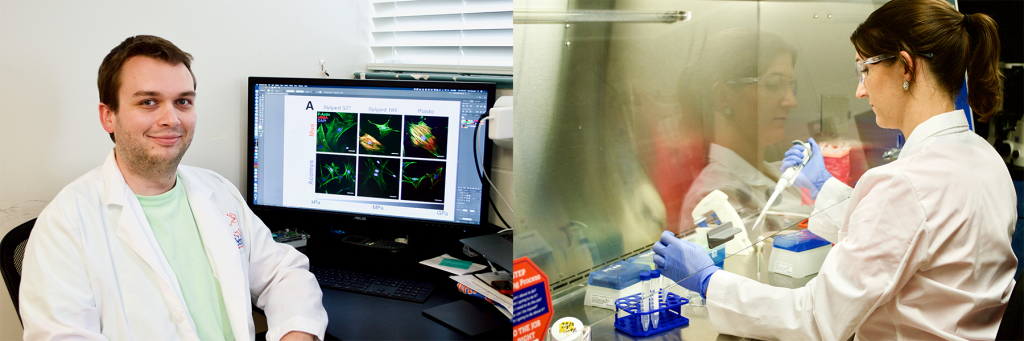
Left: Graduate student Daniel Stewart images cells to see which proteins are involved in scar-free healing.
Right: Dr. Simmons prepares cells for experiments to study Spiny Mouse regeneration.
In an earlier National Science Foundation-sponsored project, Simmons and her team looked at the ability of spiny mouse skin cells to react to their mechanical environment by stretching and contracting, and this work was recently published in the Journal of Biomechanics. In other research, scientists discovered that both the skin and the blood provide cell factors at the site of an injury that contribute to the spiny mouse’s scar-free healing. In this new project, the Simmons team will be working to transplant blood cells from the spiny mouse into other mice to see if this factor is enough to help the normal mice regenerate scar-free.
This mechanobiology project is a great example of applying engineering thinking to biology. Simmons explained, “Typical mechanical engineers think about how to design things that won’t break, how to exchange forms of energy into other forms of energy, and how to model all of these things computationally. In our project, we’re still trying to design something that doesn’t break, but in this case, it’s the human body.”
The implications for human medicine
The long-term implications for this research is significant. Scarring, or fibrosis, is present in almost every injury humans experience. Most chronic diseases and traumatic injuries result in fibrosis – kidney disease, heart attacks, spinal cord injuries, implanted devices, and more. While Dr. Simmons’ project is looking at skin scarring, the applications reach into all types of illnesses and injuries resulting in fibrosis, with the hope that someday healing is truly more than skin-deep.